Fuel Cell Cars
|
How They Work
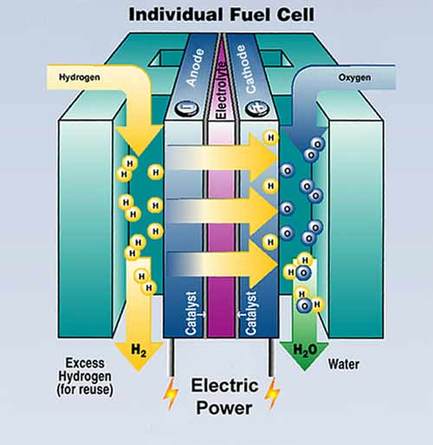
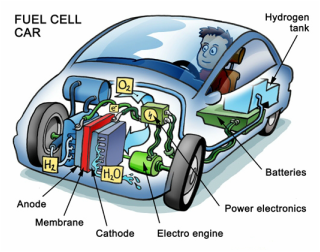
Fuel cell car batteries are similar to electric car batteries, but instead of recharging them with an electrical current the driver of the car adds more fuel for the chemical reaction. Fuel cells work by using water to form electricity. Hydrogen and oxygen are added to the car. Oxygen passes over to the cathode while hydrogen passes over to the anode with a semipermeable electrolyte medium between the two. At the anode there is a catalyst that takes away an electron from hydrogen causing hydrogen to become positively charged. When the driver of the car wants the car to run, the positive hydrogen ion can then move back to the cathode. Once there the electrons and protons are combined with oxygen to form water while they electrical current created by the passage causes the car to run. |
Efficiency
Fuel cells only have two byproducts, drinkable water and heat. This makes them much more efficient than conventional cars that produce large amounts of CO2 and NOx. A hydrogen fuel cell is much more efficient than an internal combustion engine. An internal combustion engine uses less than 20 percent of the chemical energy in gasoline. A fuel cell on the other hand uses around 60 percent of the energy available in hydrogen. Cons Fuel cell cars are most practical in isolated areas off the grid. This means that people living in cities or populated areas wouldn't have a great need for fuel cell cars. Fuel cell cars are also very expensive and harder to find than an electric or conventional car. This makes the car harder for consumers to buy and also less adaptable to everyday life. |